| Original Article | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sudan J Paed. 2023; 23(2): 214-223 SUDANESE JOURNAL OF PAEDIATRICS 2023; Vol 23, Issue No. 2 ORIGINAL ARTICLE Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell diseaseOmer S. M. Suliman (1), Hiba A. M. Elamin (2)(1) Department of Paediatrics and Child Health, Faculty of Medicine, University of Khartoum, Khartoum, Sudan (2) Paediatrics Specialist, Summerland Medical Centre, Shakhbout City, AbuDhabi, UAE Correspondence to: Omer S. M. Suliman Department of Paediatrics and Child Health, Faculty of Medicine, University of Khartoum, Khartoum, Sudan. Email: omer.suli1959 [at] gmail.com Received: 06 December 2021 | Accepted: 18 June 2023 How to cite this article: Suliman OSM, Elamin HAM. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudan J Paediatr. 2023;23(2):214–223. https://doi.org/10.24911/SJP.106-1638821311 © 2023 SUDANESE JOURNAL OF PAEDIATRICS
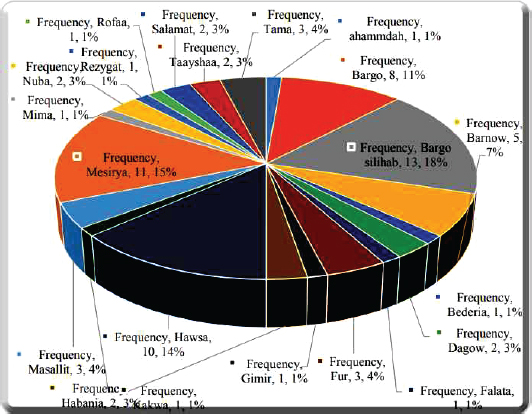
ABSTRACTA case-control interventional study was conducted to determine serum zinc levels in children with sickle cell disease (SCD) and to compare them to the levels in normal children and to the levels after 6 months of zinc supplementation. A total of 74 patients and 30 normal children, considered as controls for the zinc levels, were included. The clinical findings, including anthropometric measurements, were obtained. Serum zinc levels at the start and after 6 months, for the patients and at the start for the controls were measured. The mean age at enrolment and diagnosis were 7.5 ± 4.8 years and 5.5 ± 2.4 months, respectively. Male to female ratio was 1:1. Patients showed very low zinc levels at enrolment (0.268 ± 0.146 mg/l), while the controls had a mean zinc level at lower limits of normal (0.542 ± 0.087 mg/l) and a p-value of 0.04. After zinc supplementation, zinc levels in patients increased significantly with a p-value=0.04. Zinc supplementation had positive effects on weight and height, with a p-value of 0.001 for both. The increase in body mass index and HC were not significant, with p-values of 0.058 and 0.067, respectively. Likewise, zinc supplementation had positive effects on the haematological indices as an increase in haemoglobin levels and a decrease of leucocyte counts, with p=0.004 and 0.005, while the increase in platelet count was insignificant, p-value=0.058. Furthermore, zinc supplementation decreased the frequency of hospitalisation significantly. We recommend considering zinc supplementation as one of the standard-of-care interventions in Sudanese children with SCD. KeywordsSickle cell disease; Zinc supplementation; Hospitalisation frequency; Haematological indices. INTRODUCTIONSickle cell disease (SCD) is one of the most common inherited life-threatening disorders in humans; and predominantly affects people of African, Indian and Arab ancestry [1]. The prevalence of SCD in developed countries is increasing partly due to migration from highly prevalent countries [2]. It is now reported that over 94% of those born with SCD survive into adulthood in the US, France and the UK, in contrast to the high mortality in Sub-Saharan Africa (SSA), where 50%–90% may die in the first years of life [3]. Children with SCD are normal-sized at birth but often experience growth deficits and delayed development, which may result from chronic undernutrition, particularly zinc deficiency [4]. SCD is associated with zinc deficiency and zinc supplementation may ameliorate some of its clinical manifestations including the relief of painful crises [5]. Zinc deficiency in adult patients with SCD was first reported in 1975 [6]. Several clinical manifestations of this disorder were subsequently related to zinc deficiency. These manifestations included growth retardation, hypogonadism in males, hyperammonemia, abnormal dark visual adaptation and cell-mediated immune disorder [7]. Studies have demonstrated a significant reduction in the number of SCD-related events in people treated with zinc sulphate and also suggested a benefit for other problems in SCD, including leg ulcers, growth, infection and androgen deficiency in males [8]. The main objective of this study was to determine the status (deficiency) of zinc among children with sickle cell anaemia in comparison to normal children of similar age and sex. Other objectives were to study the effects of zinc supplementation on growth, haematological indices, including haemoglobin, total white blood cells and platelets, and the rate of admission, as a reflection of complications mainly vasoocclusive crisis (VOC) and infection, in these children. MATERIAL AND METHODSA case-control interventional study was conducted at the general wards of the Paediatric Eepartment of Ibrahim Malik Teaching Hospital, during the period from January 2019 to November 2019. Ibrahim Malik Teaching Hospital is located in Al-Sahafa city, Khartoum state. The paediatric hospital is in a separate building containing 10 general paediatric wards with a capacity of 100 beds and contains paediatric intensive care unit with 4 beds and a neonatal intensive care unit with 10 beds. It serves the whole southern area of Khartoum and also receives patients from different states of Sudan. The study population was children with SCD who were diagnosed with documented haemoglobin electrophoresis, exclusion criteria were children with SCD having chronic medical conditions, and chronic infections such as HIV and hepatitis B or C. Children with SCD who were receiving dietary supplements containing zinc were also excluded. The sample size was determined using the standard formula for a population study: N=Z2 (PQ)/D2 N=sample size, Z=statistical certainty (1.96 at 95% level of confidence), P=5% – 0.05, Q=probability of failure=1 – P=1 – 0.05=0.95 and D=desired margin of error=(0.05) or 5%. Therefore, N=1.96 × 1.96 × 0.05 × 0.95/0025=74 patients. Of these patients, 65 were admitted to the wards with different SCD crises, and the other 9 included children with SCD who were siblings of the patients called to participate in the study from home. Other 30 healthy children were selected as a control for the serum zinc level. The findings during detailed history and physical examination were entered into a comprehensive protocol sheet, which was completed for each child. The main data collected were the age of the patients, gender, age at first diagnosis with SCD, the presenting symptoms and signs at first diagnosis, tribe of origin, residency, rate of admissions and frequency of blood transfusions. Father’s and mother’s age and level of education, history of consanguinity and family history of SCD were also recorded. Complete anthropometric measurements were performed for each child at baseline and again 6 months later after zinc supplementation. Height (accurate to within 0.1 cm) was measured using a stadiometer (Holtain, Crymych, UK), each patient weighed with light clothes and without shoes to within 0.1 kg. Height, weight and calculated body mass index (BMI; weight [kg] / height [m2]) were compared to data from the newly revised WHO growth charts. Serum zinc levels were measured within 2 hours of sample collection, for the patients and control children, initially and repeated after 6 months for the 48 patients who completed the study. Serum zinc concentration (mg/l) was measured by colorimetric method using a colorimetric reagent kit ‘ACCURAS AUTO Zn’ (SHINO-TEST Corporation) for the determination of zinc in serum. During the 6-month period, all patients received a daily dose of 10 mg of elemental zinc, in the form of zinc sulphate. The data were analysed using the Statistical Package for the Social Sciences. The data were presented in the form of mean and standard deviation (SD) for quantitative variables, frequency and percentage for normal variables. The chi-squire and Student t-tests were used for the correlation between the variables and a p-value of ≤0.05 was considered significant. RESULTSA total of 74 children with SCD were included in this study. Other 30 healthy children were considered as controls for the plasma zinc levels. The mean age at enrolment was 7.5 ± 4.8 years and the mean age at the diagnosis with SCD was 5.5 ± 2.4 months. Table 1 shows the details of the age groups. Males and females were equally distributed M:F=1:1. The majority of the patients were from Khartoum state (62.2%). The mean number of admissions with painful crises was 2.0 ± 3.3 with a wide range from 0 to 20 times. The mean duration of hospitalisation was 2.1 ± 1.1 days. In this study, 55.4% of the patients received blood transfusion 1–3 times while only 8.1% were not transfused at all. All patients were receiving regular folic acid, while only 27 (36.5%) were on HU, only one patient was on oral penicillin, and seven were on omega-3 (Table 1). Most of the patients were from a second-degree consanguineous mating in 42 (56.8%); however, a considerable number were not relative to 30 (40.5%). Only 40 (54.1%) have a family history of SCD while 34 (45.9%) have no such history. About one-third of both parents are illiterate and only 8.1% of the mothers and 9.5% of the fathers had graduated from university. Table 2 shows that the most common presenting symptoms at first diagnosis were fever and pain in the upper and lower limbs and the most common signs were pallor and swelling of hands and feet (hand and foot syndrome). Almost all patients were from tribes originally from Western Sudan. The most common are the Bargo Silihab, Messeryia and Hawsa tribes (Figure 1). Patients showed very low zinc levels (mean 0.268 ± 0.146 mg/l), while the controls showed a mean zinc level at lower limits of normal (0.542 ± 0.087 mg/l) (Table 3), knowing that the normal levels of plasma zinc in children are 0.5–1.2 mg/l. The difference is statistically significant, p-value=0.04. Of the 74 patients, only 48 (64.9%) completed the study and took the zinc sulphate for 6 months which were available for comparison. Table 4 shows the correlation between patient characteristics on admission and after zinc supplementation for 6 months. The serum zinc level increased significantly from a mean of 0.265 ± 0.146 to 0.836 ± 0.471 mg/l, with a p-value=0.04. The mean increase in weight (4.6 kg) and the mean increase in height (7.61 cm) were statistically highly significant, p-value of 0.001 for each. However, the mean increase in BMI (1.1 kg/m2) and the head circumference (HC) (1.7 cm) were statistically insignificant, p-values of 0.058 and 0.067, respectively. The mean increase in haemoglobin level (0.5 g/dl) and the mean decrease in the total white blood cell count (TWBC)(8100) were statistically significant, while the increase in platelet counts was not significant (55600), p-values 0.004, 0.005 and 0.058, respectively. The frequency of admission in the patients decreased significantly after the zinc supplementation. Before the supplementation, all patients were admitted frequently to the hospital with different complications. Forty (54.1%) were admitted more than 10 times, 18 (24.3%) were admitted from 5 to 10 times, and only 16 (21.6%) were admitted less than 5 times. After the 6 months of supplementation with zinc, out of the 48 patients who completed the duration of the study, 40 (83.3%) were not admitted at all and only 8 patients (16.7%) were admitted from 1 to 2 times. Table 1. Characteristics of the patients.
SCD, Sickle cell disease. DISCUSSIONThe findings of this study showed that the mean age of the patients with SCD at diagnosis was 5.5 ± 2.5 months and about half were diagnosed at the age range between 4 and 6 months. This finding was shown by many studies [9,10] and it represents the predictable age of presentation after the natural fall of haemoglobin F (HbF) through the second haemoglobin switch from the foetal to adult haemoglobin (HbA) that occurs slowly and completed several months into postnatal period [11]. In this study, most of the patients were residing in Khartoum, although almost all patients belong to tribes from the West of Sudan, as shown by our study and a previous study [6]. This may indicate mass movements of these tribes to the capital city, Khartoum, for better medical services and for other social reasons. In the present study, a considerable number of parents were not relatives. This may indicate that the mutant gene is widely prevalent in the affected tribes. Actually, this is the exact case in most of the West African countries, where the mutant genes are highly represented. One in 4–5 (25%–30%) are carriers of HbS compared to 1/400 in African Americans [12]. In this study almost half of the mothers and fathers are illiterate. This finding was also shown by a study done in Iraq [13]. This finding might make genetic counselling difficult and a need for more efforts on family health education, which is one of the standard-of-care measures in the treatment of SCD, in addition to immunisation and penicillin prophylaxis [14]. Table 2. Symptoms and signs at first presentation.
In the current study, fever, and hand-and-foot syndrome were the most common presenting clinical features. Many studies showed similar findings, with VOC being the most common presentation [15,16]. Our reporting of frequent blood transfusion and frequent hospitalisation were also shown by many other studies in SSA [17,18]. In this study, the poor adherence to medications especially HU, may explain the high frequency of hospitalisation with infection and VOC, and the high rate of transfusion. Although numerous studies have clearly demonstrated the efficacy of HU in reducing the morbidity and mortality of SCD, a major barrier to its effectiveness has been adherence to taking this daily oral medication [19]. Two recent systematic review articles have summarised problems with adherence among paediatric patients with SCD [20,21]. The adherence rate ranged from 16% to 89%. Non-adherence with HU was associated with more VOC and hospitalisations.
Figure 1. Tribal distribution of children with sickle cell disease. In the present study, the mean serum zinc level was significantly low in children with SCD compared to healthy controls. Levels for the healthy control were in the lower limits of the normal values. This might indicate a low level of zinc in the diet of Sudanese children, which may be supported by a study done in Northern Sudan, which showed decreased zinc levels in sheep, goats, soil and plants [22]. In patients with SCD, low serum zinc levels are due to haemolysis, loss of urine and inadequate dietary intake [23]. Studies have shown that, urinary zinc loss is likely due to impaired renal tubular reabsorption of zinc [24], presumably due to tubular damage from SCD, and that bone degradation particularly during VOC leads to increased release of zinc and subsequent loss in urine [25]. Zinc deficiency in children with SCD is supported by global data showing reduced zinc levels or zinc deficiency in 15 out of 18 studies, including studies from North America (USA and Canada), South America (Brazil), the Middle East (Saudi Arabia, Turkey, Iraq and Jordan) and Africa (Nigeria and Uganda) [25–37]. Table 3. Mean serum zinc levels of the patients and the control.
Normal plasma zinc level 0.5–1.2 mg/l; p-value 0.04. This study showed significant effects of zinc supplementation on serum zinc levels, rate of hospitalisation, growth parameters and haematological indices. The serum zinc level increased significantly. This effect has been shown by many other studies [38–40]. These results provide clear evidence that a simple regimen of oral zinc supplementation corrects zinc deficiency. The hospitalisation rate decreased significantly after the zinc supplement. The effects of zinc supplementation on decreasing the incidence of VOC, infection and transfusion rates may explain this reduction in hospitalisation. These effects were shown by many other studies [41,42]. In our study, zinc supplementation has significant effects on height and weight. However, the increase in BMI and HC were not significant. The positive effects of zinc supplementation on growth have been shown by many other studies [43–45]. The low Hb in SCD patients is obviously due to haemolysis caused by the genetic mutation of Hb, and hence one should not expect the Hb level to increase following zinc supplementation. However, zinc facilitates red cell deformability, so that in the case of SCD, there is a reduction of red cells destroyed by the spleen, so may increase Hb levels [1]. Table 4. Comparison between the patients’ characteristics on admission and after zinc supplements.
*Values are given as mean ± SD. BMI, Body mass index; Hb, haemoglobin; HC, head circumference; TWBC, total white blood count. The function of neutrophils in promoting vaso-occlusive events (VOEs), is documented in many reports. The severity of clinical manifestations of SCD has been associated with leucocytosis and neutrophilia in one study [46]. A recent study also demonstrated that micro-vascular oxygen consumption and neutrophil counts are further elevated in SCD patients during VOE [47]. Leucocytosis also positively correlates with early death, silent brain infracts, haemorrhagic strokes and acute chest syndrome in SCD patients [48], suggesting the critical role of leucocytes, and neutrophils in particular, in SCD morbidity and mortality. Different from neutrophils, which further increase before the onset of VOE, platelet count is high at steady state and declines during acute VOE [49]. Limitations of the study consist of the following:
CONCLUSIONIn the present study, children with SCD had frequent hospitalisation with complications, low growth parameters and poor adherence to medications, especially HU. Furthermore, they had low serum zinc levels, compared to healthy children, who, themselves had marginal zinc deficiency, suggesting dietary zinc deficiency in Sudanese children. Zinc supplementation had positive effects on serum zinc levels, growth parameters especially weight and height and in reducing hospitalisation rates, possibly by decreasing infection and VOC. In addition, zinc had positive effects on the haematological indices by increasing Hb levels and decreasing TWBC. We recommend considering zinc supplementation as one of the standard-of-care interventions in Sudanese children with SCD. CONFLICT OF INTERESTThe authors declare that they have no competing interests. FUNDINGNone. ETHICAL APPROVALThe study was approved by the Sudan Medical Specialisation Board. An informed written consent was obtained from the parents of the patients and the control. REFERENCES
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| How to Cite this Article |
| Pubmed Style Suliman OSM, Elamin HAM. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudan J Paed. 2023; 23(2): 214-223. doi:10.24911/SJP.106-1638821311 Web Style Suliman OSM, Elamin HAM. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. https://sudanjp.com//?mno=40145 [Access: July 05, 2025]. doi:10.24911/SJP.106-1638821311 AMA (American Medical Association) Style Suliman OSM, Elamin HAM. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudan J Paed. 2023; 23(2): 214-223. doi:10.24911/SJP.106-1638821311 Vancouver/ICMJE Style Suliman OSM, Elamin HAM. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudan J Paed. (2023), [cited July 05, 2025]; 23(2): 214-223. doi:10.24911/SJP.106-1638821311 Harvard Style Suliman, O. S. M. & Elamin, . H. A. M. (2023) Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudan J Paed, 23 (2), 214-223. doi:10.24911/SJP.106-1638821311 Turabian Style Suliman, Omer S M, and Hiba A M Elamin. 2023. Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudanese Journal of Paediatrics, 23 (2), 214-223. doi:10.24911/SJP.106-1638821311 Chicago Style Suliman, Omer S M, and Hiba A M Elamin. "Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease." Sudanese Journal of Paediatrics 23 (2023), 214-223. doi:10.24911/SJP.106-1638821311 MLA (The Modern Language Association) Style Suliman, Omer S M, and Hiba A M Elamin. "Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease." Sudanese Journal of Paediatrics 23.2 (2023), 214-223. Print. doi:10.24911/SJP.106-1638821311 APA (American Psychological Association) Style Suliman, O. S. M. & Elamin, . H. A. M. (2023) Zinc level and effect of zinc supplementation on growth in a subset of Sudanese children with sickle cell disease. Sudanese Journal of Paediatrics, 23 (2), 214-223. doi:10.24911/SJP.106-1638821311 |
Nagwa Salih, Ishag Eisa, Daresalam Ishag, Intisar Ibrahim, Sulafa Ali
Sudan J Paed. 2018; 18(1): 24-27
» Abstract » doi: 10.24911/SJP.2018.1.4
Siba Prosad Paul, Emily Natasha Kirkham, Katherine Amy Hawton, Paul Anthony Mannix
Sudan J Paed. 2018; 18(2): 5-14
» Abstract » doi: 10.24911/SJP.106-1519511375
Inaam Noureldyme Mohammed, Soad Abdalaziz Suliman, Maha A Elseed, Ahlam Abdalrhman Hamed, Mohamed Osman Babiker, Shaimaa Osman Taha
Sudan J Paed. 2018; 18(1): 48-56
» Abstract » doi: 10.24911/SJP.2018.1.7
Adnan Mahmmood Usmani; Sultan Ayoub Meo
Sudan J Paed. 2011; 11(1): 6-7
» Abstract
Mustafa Abdalla M. Salih, Mohammed Osman Swar
Sudan J Paed. 2018; 18(1): 2-5
» Abstract » doi: 10.24911/SJP.2018.1.1
Amir Babiker, Afnan Alawi, Mohsen Al Atawi, Ibrahim Al Alwan
Sudan J Paed. 2020; 20(1): 13-19
» Abstract » doi: 10.24911/SJP.106-1587138942
Hafsa Raheel, Shabana Tharkar
Sudan J Paed. 2018; 18(1): 28-38
» Abstract » doi: 10.24911/SJP.2018.1.5
Anita Mehta, Arvind Kumar Rathi, Komal Prasad Kushwaha, Abhishek Singh
Sudan J Paed. 2018; 18(1): 39-47
» Abstract » doi: 10.24911/SJP.2018.1.6
Majid Alfadhel, Amir Babiker
Sudan J Paed. 2018; 18(1): 10-23
» Abstract » doi: 10.24911/SJP.2018.1.3
Bashir Abdrhman Bashir, Suhair Abdrahim Othman
Sudan J Paed. 2019; 19(2): 81-83
» Abstract » doi: 10.24911/SJP.106-1566075225
Amir Babiker, Mohammed Al Dubayee
Sudan J Paed. 2017; 17(2): 11-20
» Abstract » doi: 10.24911/SJP.2017.2.12
Cited : 8 times [Click to see citing articles]
Mustafa Abdalla M Salih; Satti Abdelrahim Satti
Sudan J Paed. 2011; 11(2): 4-5
» Abstract
Cited : 4 times [Click to see citing articles]
Hasan Awadalla Hashim, Eltigani Mohamed Ahmed Ali
Sudan J Paed. 2017; 17(2): 35-41
» Abstract » doi: 10.24911/SJP.2017.2.4
Cited : 4 times [Click to see citing articles]
Amir Babiker, Afnan Alawi, Mohsen Al Atawi, Ibrahim Al Alwan
Sudan J Paed. 2020; 20(1): 13-19
» Abstract » doi: 10.24911/SJP.106-1587138942
Cited : 4 times [Click to see citing articles]
Mutasim I. Khalil, Mustafa A. Salih, Ali A. Mustafa
Sudan J Paed. 2020; 20(1): 10-12
» Abstract » doi: 10.24911/SJP.1061585398078
Cited : 4 times [Click to see citing articles]